| Author |
Topic: continuation of Wizard x's "One Step"
thread |
dwarfer
Member |
 posted 09-20-1999 03:49 PM
posted 09-20-1999 03:49 PM

I tried to post to the referenced thread but it would accept no more
messages.
??
Anyway, my understanding of the bubbling of the H2S is to generate SO3
radical.
H2S has its share of hazards in use and smell.
COULD Na thiosulfate, which generates
.....S2O3->SO3 + S.......... be produced IN the mixture through
gradual acidification, or, perhaps through electrolytic means?
If so, and if pressurization would be an asset to the effectiveness of
the process, this could be the cat's meow, or the molasses' revenge:
easy for me to get excited: in my mind it works perfectly, being
unconstrained by nuisance considerations of chemical minutia.
Might need a particular anode: might need a 1/2 cell: might need good
agitation.
Might be ridiculous.
I oughta change my name to "slap me if I need it" for these far out
what if posts.
dwarfer
|
dwarfer
Member |
 posted 09-20-1999 04:02 PM
posted 09-20-1999 04:02 PM

Here's some names I tried to open that were "allready taken"!! (Hard to
believe!)
"OK, I'm a dumb shit"
"Slap me if I need it"
"Not too smart but I
try"
"??but I try??
"Comeon: have some pity!!"
Oh well: I tried 
|
Hematite
Member |
 posted 09-21-1999 12:11 PM
posted 09-21-1999 12:11 PM

Dwarfer,
I know exactly what you mean. It always makes me laugh at how
we attach so many of our human emotions to our deliberately invented
anonymous posting names.
Ps. If your real name is Dwarfer, then
Hematite most humbly apologises.
PPs. Apologises for your parents
cruelty that is 
------------------
Regards,Hematite.
|
dwarfer
Member |
 posted 09-21-1999 08:01 PM
posted 09-21-1999 08:01 PM

Yeah, it's a surprise to me to note the attachment you get after a while
to a stupid name. i made up dwarfer on the spur of the minute, since I WAS
posting under my name when I first started: soon realized that was not
tooooooooooooooo smart, so went to register and "dwarfer" popped up.
Hell, if I thought about it I woulda picked "Petunia" or something
sweet and nice like I am, really!
===================
Now if I could JUST get Wiz or Rev. to comment on the thiosulphate
idea.
I'm willing to experiment if the idea is not TOO ludicrous: but do not
want to waste my time if its obviously a non-runner.
hey THHHHHHpitterdude(dodge the spray): howabout using your not
inconsiderable influence with the Reverend to inveigle him into
critiqueing this possibility??
TTTHHHHHHanx!
dwarfer
|
Overlord
Junior
Member |
 posted 09-21-1999 09:44 PM
posted 09-21-1999 09:44 PM

bad wiz... you are bad 
|
Wizard
X
Member |
 posted 09-23-1999 02:37 AM
posted 09-23-1999 02:37 AM

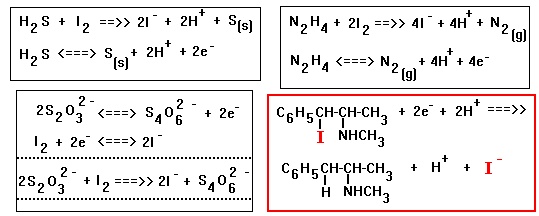
The quantitative conversion of the thiosulphate, S2O3(-2) ion to
tetrathionate, S4O6(-2) ion is unique with iodine, I2; OTHER oxidizing
agents Cl2 and Br2, tend to carry the oxidation further, to sulphate ions
or a mixture of sulphate ions and tetrathionate ions.
The thiosulphate, S2O3(-2) ion tend to decompose to give sulphur and
hydrogen sulphite ion: S2O3(-2) + H(+) <==> HSO3(-) + S
The rate
of the decompostion reaction increases markedly with increasing acidity.
When thiosulphate, S2O3(-2) is added to a strong acidic solution, the
decompostion is immediate and the solution turns cloudy due to sulphur
precipitating out.
I believe addition of thiosulphate, S2O3(-2) to iodoephedrine will
result in dimerization of the iodoephedrine, due to radical formation and
coupling. The thiosulphate, S2O3(-2) will release 2 electrons, BUT NO
protons H(+) to react with the I(-) ion formed and the
C6H5-CH(..)-CH(NHCH3)-CH3 radical anion formed.
Hydrogen sulphide, H2S and Hydrazine, N2H4 will release electrons and
protons for the reduction of iodoephedrine.
C6H5-CH(I)-CH(NHCH3)-CH3 + H2S ==>> C6H5-CH2-CH(NHCH3)-CH3 + S +
HI
2 C6H5-CH(I)-CH(NHCH3)-CH3 + N2H4 ==>>
2
C6H5-CH2-CH(NHCH3)-CH3 + N2 + 2HI
|
Worlock
Member |
 posted 09-23-1999 03:02 PM
posted 09-23-1999 03:02 PM

The Sodium thiosulfate does seem to clear the redness from the final
acidic solution in the HI/RP solution because as the Sodium thiosulfate
hits the acid it initially will ionize into Na+ and thiosulfate.
The
Na+ and the I- form [Na+ I-]
which eliminates the color.
Then as Wizard-x notes in detail the thiosulfate degenerates.
Once again then I return to the question of why use Sodium thiosulfate
to clear the iodide out of the finished reaction mixture.
Because as soon as the alkalizing begins with Sodium hydroxide the same
thing happens the Na+ ionizes then hooks up with I- to form
NaI.
To cap this off with my final thrust at I2, realise that in an Acidic
Solution the existance of I2 is highly dubious.
|
Wizard
X
Member |
 posted 09-23-1999 07:25 PM
posted 09-23-1999 07:25 PM

Worlock : "Once again then I return to the question of why use Sodium
thiosulfate to clear the iodide out of the finished reaction
mixture".
To convert the I2 into a soluble ion I(-) so you do not
solvent extract I2 when you extract your final product.
|
Fitzhugh
Member |
 posted 10-10-1999 04:47 AM
posted 10-10-1999 04:47 AM

Wizard X
The way I read the hydrazine reaction would mean that I would add one
half of a mole of N2H4 for every mole of I2 to produce the initial HI, and
I would add one-half of a mole of N2H4 for every mole of p/efed to remove
the I and replace it with an H, and regenerate the HI.
This process looks like it would generate H2O as well.
Have you a suggestion for actually running this reaction?
Combine ingredients in the proper ratios with a minimum of water and
reflux for a long time? A short time?
This sounds too interesting to ignore.
|
Wizard
X
Member |
 posted 10-10-1999 07:40 PM
posted 10-10-1999 07:40 PM

If you want to make iodoephedrine first you add 1 mole of hydrazine N2H4 :
4 moles of iodine to make 4 moles of HI, and then you add 2 moles of
ephedrine. Refluxing this for approx 24 hours gives iodoephedrine.
Then
2 moles of iodoephedrine : 1.1 moles of hydrazine.
The other method is
to reduce 1 moles of ephedrine with 2 moles of hydrazine.
|
Fitzhugh
Member |
 posted 10-14-1999 07:30 AM
posted 10-14-1999 07:30 AM

If one were willing to dream of testing some variations of the E + N2H4
method you mention, would you have any suggestions for initial conditions,
reaction temperatures or durations? (I know that N2H4 reacts more strongly
at higher temperatures.)
The generation on N2 gas would be an indicator of the reaction's
progress.
-- Fitzhugh--
|
whoops
Member |
 posted 11-13-1999 05:11 PM
posted 11-13-1999 05:11 PM

once again rephrase the method or question?
Dont everybody understand the removal point being considered by making
the freebase ya are removing the Haloid from the mixture and it is gona co
[do not miss this is not a typo idiot]Carbon monoxide is formed from the
classical, or shall we also consider the solvent burning or well we could
complicate this further by discussing the phosphor,,,NO THANKS  solvate
with a strong base such as lye in the water layer the lesser base the
amine will swim into the solvent where there is alot of hydrogen trapped
in the matrix, or lattice therin, the free base of it should be void of at
least one thing, the haloid interaction, wheater or not this problem of
the ripping of the oxygen of the molecule can be preformed another way is
a different and excitting idea, but it seems there are simpler ways, thus
if ussing Iodine ya can form a salt simmiliar to HCL and thus to it can
also form HI and what ya are hopping to do is twist off one of the oxygen
atoms containing the oh group, the halid is capable of this under
increased tempature and friction is ussed in these classical therums to
rip the thing away, Now how this happens is a matter of theory, the best
explainations are formed from metal addation in my opinion solvate
with a strong base such as lye in the water layer the lesser base the
amine will swim into the solvent where there is alot of hydrogen trapped
in the matrix, or lattice therin, the free base of it should be void of at
least one thing, the haloid interaction, wheater or not this problem of
the ripping of the oxygen of the molecule can be preformed another way is
a different and excitting idea, but it seems there are simpler ways, thus
if ussing Iodine ya can form a salt simmiliar to HCL and thus to it can
also form HI and what ya are hopping to do is twist off one of the oxygen
atoms containing the oh group, the halid is capable of this under
increased tempature and friction is ussed in these classical therums to
rip the thing away, Now how this happens is a matter of theory, the best
explainations are formed from metal addation in my opinion  such
as that sulfer is combined to the oxygen atom in a cascade that eventually
rips way too the activly transiting site, such that other metals are often
employed such as lithium or aluminium or sodium, any of these metals
considering whatever reaction might be employed, the real question I find
paculiar about HI is this that the final critique ends up being as from
florine working your way through the respective haloids bromine clorine
and finally the iodine has the most metaloid like properties and therefore
is a catalys that preforms, it is the not so preffered catalyst by any
stretch of the immagination it just works very well under a great varity
of conditions and this is good for begeners. Now someone out there does
know an easy and gentle way to be shure and the final work up of the
product so happens to conclude with a shared acid bond formation
paticuliar to the subject at hand being discussed for educational
purposes, thus the sulferuos conponent is a shared acid that the isomeric
amines share between them, this results in dont trust the litature it aint
always right nor will it ever be fixed, hum war inside your head, nothing
to be scared of except your imagination in any case.....hummm so the
molecule is at an odd charge is it??? the sharring of from the two results
in an odd charge dont it???? such
as that sulfer is combined to the oxygen atom in a cascade that eventually
rips way too the activly transiting site, such that other metals are often
employed such as lithium or aluminium or sodium, any of these metals
considering whatever reaction might be employed, the real question I find
paculiar about HI is this that the final critique ends up being as from
florine working your way through the respective haloids bromine clorine
and finally the iodine has the most metaloid like properties and therefore
is a catalys that preforms, it is the not so preffered catalyst by any
stretch of the immagination it just works very well under a great varity
of conditions and this is good for begeners. Now someone out there does
know an easy and gentle way to be shure and the final work up of the
product so happens to conclude with a shared acid bond formation
paticuliar to the subject at hand being discussed for educational
purposes, thus the sulferuos conponent is a shared acid that the isomeric
amines share between them, this results in dont trust the litature it aint
always right nor will it ever be fixed, hum war inside your head, nothing
to be scared of except your imagination in any case.....hummm so the
molecule is at an odd charge is it??? the sharring of from the two results
in an odd charge dont it????
IT AN ODD CHARGE AINT IT???????
---Amethystium---
|

 the
Hive BB
the
Hive BB 
 Crystal
Meth
Crystal
Meth 
 continuation
of Wizard x's "One Step" thread
continuation
of Wizard x's "One Step" thread




